Ahmed wrote:...and the worst part is that its origin has absolutely no importance!

Great on the contrary for scientists. I don't understand why interest escapes you

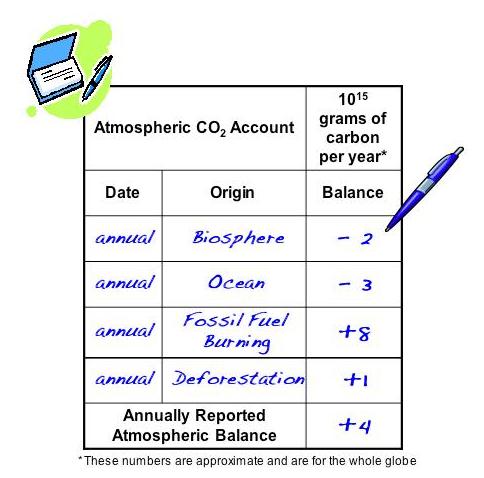
This allows researchers, by analyzing an air sample, with a mass spectrometer, to determine the origin of the CO2 contained and thus be able to demonstrate the fossil and "natural" part, table that I put a little above

And many other inventories
https://gml.noaa.gov/outreach/isotopes/c14tellsus.htmlThe history of CO2
https://gml.noaa.gov/outreach/isotopes/mixing.htmlLocal air pollution status
https://gml.noaa.gov/outreach/isotopes/sampling.htmlext...
The relative proportion of 13C in our atmosphere is steadily decreasing over time. Before the industrial revolution, δ13The C of our atmosphere was around -6,5‰; Now the value is around -8‰. Remember that plants have minus 13C relative to the atmosphere (and therefore have a more negative δ13C of around -25‰). Most fossil fuels, such as oil and coal, which are ancient plant and animal materials, have the same δ13C isotopic as other plants. The annual trend, i.e. the overall decrease in atmospheric δ13C– is explained by the addition of carbon dioxide to the atmosphere which must come from the terrestrial biosphere and/or fossil fuels. In fact, we know from Δ14C measurements, inventories and other sources, that this decrease is due to fossil fuel emissions, and is an example of the Suess effect.
https://gml.noaa.gov/outreach/isotopes/c13tellsus.html