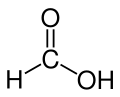
Storing hydrogen by using Co2 to make formic acid, in order to more easily produce and store energy in the form of “chemically encapsulated hydrogen”, this is the feat achieved by EPFL researchers. This is the discovery of the Grail, since the return of hydrogen no longer presents a danger ... No, we are not on April 1: we will not see the 100% water engine, but the formic acid engine (HCOOH or CH2O2) yes, most certainly.
The end of the greenhouse and nuclear nightmare
This discovery thus makes it possible to convert thermal energy / electricity into a “fuel”, then by the reverse process to recover electricity or heat. With the decisive advantage, that it is not dangerous to store like hydrogen was in pressurized cylinders. In addition, formic acid has a "Flash point" has a much higher temperature than gasoline.

Electricity from a wind turbine or PV panels can therefore be stored in the form of formic acid (by capturing the passage of Co2, which we are trying to get rid of ...) probably in the form of a battery , or even available in jerrycan (two packaging evoked by EPFL).
So, when there is no sun or no wind, we can use formic acid by releasing the hydrogen it contains as an energy source! Different scenarios are then possible to store transport produce heat and / or electricity.
Wind, offshore solar plants / deserts, etc.: a new rational use of solar
The system presented can be used both in a car and for an individual installation, in a house, etc.

Almost unnoticed a few months before Fukushima, here is a discovery which must have weighed heavily in the decision of the respective Swiss and German governments to leave nuclear power! And that is bound to change the world:
Storage of hydrogen via formic acid
Ideal solution to accumulate energy from renewable sources such as solar or wind, and also power the car of tomorrow.
Announced yield> 60%
Video:
Or:
Full text of EPFL, Alain Herzog © 01.12.10 wrote:Formic acid in the engine
Catalysis producing hydrogen from formic acid
Do ants hold the key to the fuel of the future? Formic acid allows more efficient and secure storage of hydrogen. An ideal way to accumulate energy from renewable sources or propel the car of the XNUMXst century.
Hydrogen is often referred to as the future alternative to fossil fuels. Ecological and efficient, it nevertheless has many drawbacks. Extremely flammable, it must be stored in bulky pressurized bottles. So many obstacles to its use, that EPFL scientists and their colleagues from Leibniz-Institut für Katalyse have removed: once transformed into formic acid, hydrogen can be stored easily and safely. An ideal solution to accumulate energy from renewable sources such as solar or wind, or power the car of tomorrow.
Hydrogen is easily produced from electrical energy. Thanks to a catalyst and CO2 present in the atmosphere, scientists have transformed it into formic acid. Rather than a heavy cast iron bottle filled with hydrogen under pressure, they thus obtain a substance with very low flammability and liquid at room temperature.
In November 2010, second step. The EPFL laboratories have managed to provoke the opposite phenomenon: through catalysis, formic acid returns to the state of CO2 and hydrogen, which can then be transformed into electrical energy. A functional, space-saving prototype with a power of 2 kilowatts is already developed. Two companies have purchased a license to develop this technology: Granit (Switzerland) and Tekion (Canada).
Store renewable energy
"Imagine for example that you have solar cells on your roof," explains Gabor Laurenczy, professor at the Laboratory of Organometallic and Medicinal Chemistry and head of Catalysis Group for Energy and the Environment. In bad weather or overnight, your formic acid battery gives you back the overflow of energy accumulated when the sun was shining. ”In such a configuration, the process makes it possible to restore more than 60% of the electrical energy of departure.
This solution is extremely safe. Formic acid continuously releases very small amounts of hydrogen, "just what you need right now for your electricity consumption," notes the researcher.
Another advantage compared to conventional storage, the process makes it possible to store almost twice the energy at equal volume. Indeed, a liter of formic acid contains more than 53 grams of hydrogen, against barely 28 grams for the same volume of pure hydrogen pressurized at 350 bars.
researchers worked on an iron-based catalysis process - a readily available and inexpensive metal, in comparison with "noble" metals such as platinum or ruthenium. As in all catalysts, no material is degraded during the process.
Formic acid at the pump
It is undoubtedly in the automotive field that the invention presents the most interesting potentials. Currently, the prototypes produced by certain major brands store hydrogen in conventional form, with the problems that we know: danger of explosion, large volume occupied by the pressurized tank, difficulties to refuel quickly ...
2008st century vehicles could run on formic acid. This solution makes hydrogen storage not only safer, but also more compact and easier to fill at the pump - formic acid is liquid at room temperature. “Technically, it is entirely doable. Moreover, major manufacturers contacted us in XNUMX, when the barrel of oil reached peaks, says Gabor Laurenczy. In my opinion, the only obstacle is economic. ”It will take a few more years before perhaps being able to refuel at the first anthill crossed on the way.
Source: http://actu.epfl.ch/news/formic-acid-in-the-engine/
[EDIT: m-à-j] A first approach had been made, notably by the Germans, with Professor Dr. Arno Behr of the faculty of chemical and biological engineering in Dortmund, who was already talking about the capture of Co2, in a process involving formic acid:
https://www.econologie.com/des-scientif ... -1243.html