At equal engine efficiency, ethanol thus emits 4,56 / 3,55 = 1,28 times more CO2 than gasoline to provide the same work.
In other words: ethanol emet, 28% CO2 in addition to gasoline.
And are not included in this calculation for ethanol:
- the CO2 production costs of the cereal or sugar plant
- the costs of refining the plant in sugar
- the costs of distillation
- energy costs of fermentation (ie the CO2 emitted to heat the molasses)
Some reminders of chemistry: sugar, fermentation, alcohol and CO2 ...
We will retain the following atomic masses which serve as the basis of all the calculations: C = 12 g / mol, O = 16 g / mol, H = 1 g / mol
a) The case of alcohol resulting from the fermentation of sugar
The sugar used to make ethanol is from the family of diholosides (sucrose or maltose) of formula: C12H22O11 (see http://fr.wikipedia.org/wiki/Sucre#Chimie ) has a molar mass of 342 grams which, by hydrolyzing (it "gains" a molecule of water), will give 2 molecules of glucose or 2 * C6H12O6 with a molar mass of 180 grams for a total mass of 360 grams.
Glucose Details: http://fr.wikipedia.org/wiki/Glucose
The mole of glucose will then ferment according to the equation:
code: Select all
C6H12O6 --> 2*C2H6O + 2*CO2Products: 2 moles of ethyl alcohol (C2H6O = 46g / mol) and two moles of carbon dioxide (CO2 = 44g / mol).
Before being burned and neglecting any other CO2 emissions, each gram of alcohol produced will therefore release, during its "creation": 44/46 = 0.95 gram of CO2.
And this BEFORE its combustion. During the combustion of alcohol:
code: Select all
C2H6O + 7/2*O2 --> 2*CO2 + 3*H2O or for those who prefer round numbers:
code: Select all
2*C2H6O + 7*O2 --> 4*CO2 + 6*H20Balancing error on the reagents, rectifying (which does not change the results on the CO2):
code: Select all
C2H6O + 3*O2 --> 2*CO2 + 3*H2O While burning, this mole of 46 g of alcohol will therefore produce 88 g of CO2 (2 moles).
Thus, a total of 3 moles of CO2 emitted per mole of burned alcohol: 1 during the fermentation and 2 during the combustion is obtained. In total 3x44 = 132 g of CO2 for 46 grams of burned alcohol.
Conclusion: each gram of alcohol thus emits during its elaboration and combustion at least 132 / 46 = 2,87 grams of CO2.
I say at least because many "CO2 positions" have not been taken into consideration: sugar production and alcohol distillation are undoubtedly the most energy-hungry.
This value of 2,87 g CO2 / g of alcohol is therefore a very minor value!
b) The case of gasoline (which alcohol is supposed to replace)
I resume without detailing the calculations of this page: combustion equations and CO2
Emissions of CO2 for a hydrocarbon of formula C2H (2n + 2) = 44n
Application to gasoline (pure octane). n = 8
[C8H18] = 8 12 * + = 18 1 114 * g / mol.
The mass of CO2 released per mole of octane consumed is: 44 * 8 = 352 g.
The fuel consumption ratio on CO2 releases is 352 / 114 = 3,09
Conclusion: each gram of gasoline emits during its combustion 3,09 grams of CO2
The cost of "refining" being, at worst, 15%, one would obtain 3.55 g CO2 for the refining and the combustion of gasoline.
c) Ethanol and gasoline energy comparison
code: Select all
PCI ethanol = 26 900 kJ/kg
PCI essence = 42 700 kJ/kg
At engine speed, 42,7 / 26,9 = more ethanol mass is required to provide the same energy, ie 1,59 times.
In isoenergy, it is therefore necessary to add a coefficient of 1,59 to the CO2 emissions of ethanol: the 2,87 grams thus become 4,56 grams. And remember: these 2,87 g were already largely minus because they did not take into account the sugar culture (but skeptics will say, with reason, that for gasoline, we did not take into account the cost of extraction and transport of crude)
General conclusion
To provide the same amount of combustion energy, the CO2 emissions are respectively:
- with gasoline: 3,55 grams of CO2
- with ethanol: 4,56 grams of CO2
At equal engine efficiency, ethanol thus emits 4,56 / 3,55 = 1,28 times more CO2 than gasoline to provide the same work.
In other words: ethanol emet, 28% CO2 in addition to gasoline.
And are not included in this calculation for ethanol:
- the CO2 production costs of the cereal or sugar plant
- the costs of refining the plant in sugar
- the costs of distillation
- energy costs of fermentation (ie the CO2 emitted to heat the molasses)
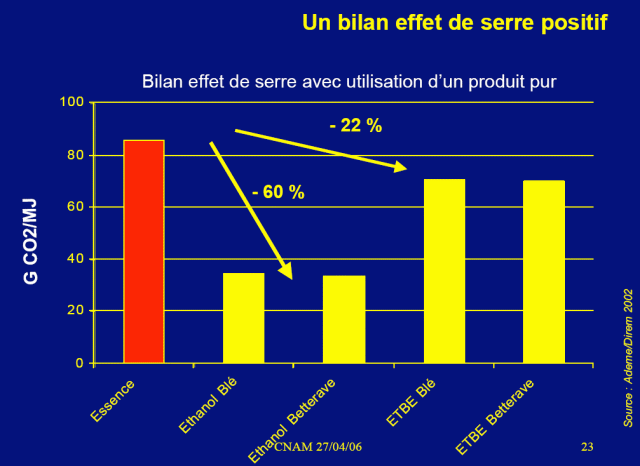
There is therefore no doubt that, based on this reasoning, ethanol of agricultural origin emits much more CO2 than gasoline. So how can state agencies and universities get CO2 reviews on ethanol so positive? Do not they take into account fermentation? The omissions are they? Or where is the flaw in our reasoning?